Chinese scientists from Institute of Solid State Physics, Hefei Institutes of Physical Science developed a green biomass electrocatalytic conversion system.
Their work not only gives guiding significance to designing both highly active catalyst for electrocatalytic hydrogenation (ECH) and electrocatalytic synthesis system.
Biomass, as an important product of green plants photosynthesis, has been regarded as an important alternative to fossil energy to produce fuels and chemicals.
In light of the large production and wild distribution, lignocellulosic biomass have raised concern in recent years, and the utilization of lignocellulose not only avoid the previous problems that biofuels from food would compete with people about food, and compete with food about the land, also contribute to the environment protection and make the waste into treasure.
However, the fine chemicals manufacturing from biomass derived platform molecules represent a challenging issue because of limited conversion techniques, harsh reaction conditions, poor catalytic performance and product selectivity, complex reaction systems and reaction mechanism.
For example, the conventional biomass valorization is often accomplished through thermocatalytic conversion processes with high pressure H2 or O2 at high temperature and high pressure conditions employing all kinds of heterogeneous catalysts, usually containing noble metals.
In this new work, the researchers took lignocellulosic biomass derived furfural upgrading as a concept proof model, simultaneously realized high-rate furfural electrocatalytic hydrogenation and oxidation upgrading on self-designed transition metal phosphide electrodes at ambient conditions.
Through the D2O isotope labeling method, they, for the first time, directly proved that the hydrogen for ECH of furfural was the hydrogen from water.
By using DFT method, the competition mechanism between ECH and H2 evolution from water has been completely studied.
This work was supported by the Natural Science Foundation of China (Grant No. 51672277 and 51432009), the CAS Pioneer Hundred Talents Program, and the CAS/SAFEA International Partnership Program for Creative Research Teams of Chinese Academy of Sciences, China.
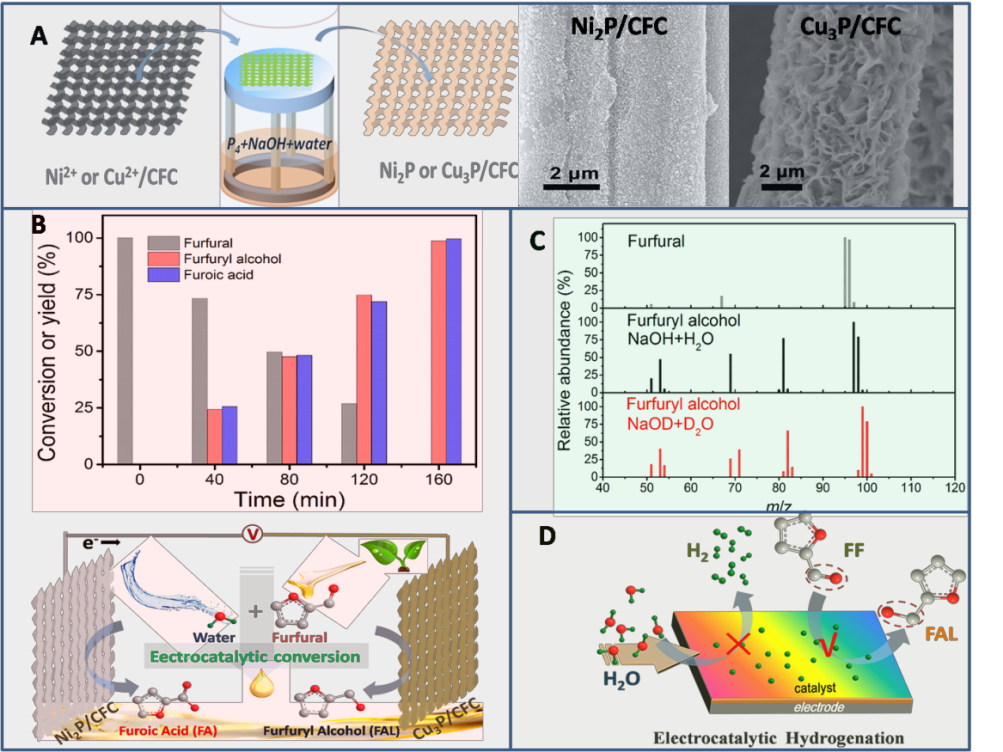
Figure (A) A schematic illustration of the fabrication process, and the SEM images of as-prepared electrodes; (B) Conversion and yield (%) changes of furfural and its oxidation/hydrogenation products, and schematic diagram of two-electrode electrocatalytic conversion of furfural. (C) Mass spectra of furfural and its products obtained in H2O/NaOH and D2O/NaOH. (D) The schematics illustrating the ECH and HER at catalyst surface.(Image by ZHANG Xian)
Contact:
ZHOU Shu
Hefei Institutes of Physical Science (http://english.hf.cas.cn/)
Email: zhous@hfcas.ac.cn
 Tel: +86-551-65591206
Tel: +86-551-65591206
 Fax: +86-551-65591270
Fax: +86-551-65591270
 Emai: zhous@hfcas.ac.cn
Emai: zhous@hfcas.ac.cn
 350 Shushanhu Road
350 Shushanhu Road