
A research team including XU Wan, LIU Xiao-Di and Eugene Gregoryanz from Hefei Institute of Solid-state Physics (ISSP), Hefei Institutes of Physical Science (HFIPS) led the international collaboration consisting of the School of Physics, University of Edinburgh, UK and HPSTAR, Shanghai, China to study the spontaneous ionization of Ammonia hemihydrate (AHH) under compression. They discovered Ionic Phases of Ammonia-Rich Hydrate at High Densities and the results of this study were recently published in Physical Review Letters.
Mixtures of ammonia and water are major components of the “hot ice" mantle regions of icy planets. The ammonia-rich ammonia hemihydrate (AHH) plays a pivotal role as it precipitates from water-rich mixtures under pressure.
The team compressed an exemplary ammonia-water compound to over 1.2 million atmospheres, corresponding to a depth of ~9870/8085 km inside Uranus/Neptune, and tracked its structural and dynamical properties. The calculations done by the team demonstrated that the compressed mixture transforms into a very stable ammonium oxide, (NH4+)2O2-.
The presence of such a stable ionic compound deep inside icy planets will influence our understanding about such planets’ formation and evolution to the present day. These intriguing ionic phases can exist over a considerably wide pressure region hence greatly extend the phase diagram of AHH.
This work was supported by National Natural Science Foundation of China, CAS Innovation Grant, Science Challenge Project, and the HFIPS Director's Fund et al.
Link to the paper: Ionic Phases of Ammonia-Rich Hydrate at High Densities
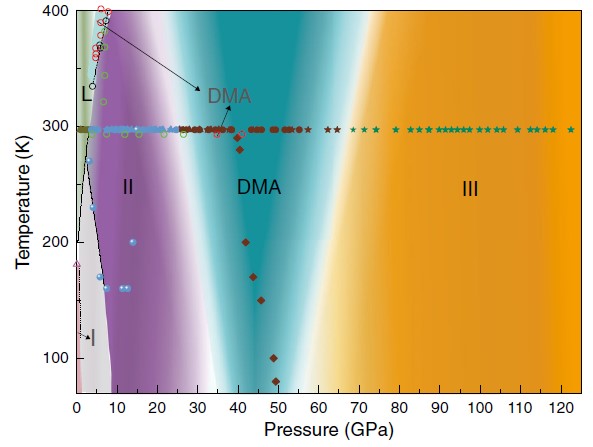
The proposed phase diagram of AHH based upon this Letter and low-pressure data from Wilson et al. For the experimental data, different colors depict different phases, and different filled symbols represent different runs. (Image by XU Wan)
Contact:
ZHAO Weiwei
Hefei Institutes of Physical Science (http://english.hf.cas.cn/)
Email: annyzhao@ipp.ac.cn