
A research group led by Prof. ZANG Haimin from the Institute of Solid State Physics, Hefei Institutes of Physical Science reported their finding about inactivation and regeneration of highly ordered Nb2O5 nanochannel film catalyst for electrocatalytic N2 reduction.
Ammonia (NH3) has been widely applied in fuel of vehicles, agricultural, plastic and textile industries. Its production from N2 and H2 release a large amount of CO2. Electrocatalytic N2 reduction reaction (NRR) at ambient condition has been regarded as a promising NH3 synthesis method for replacing the traditional energy- and capital-intensive Haber-Bosch process.
"Our goal is to develop an efficient and economic catalyst for NRR", said Prof. ZHANG Haimin, a scientist of chemistry, “so we fabricated highly ordered Nb2O5 nanochannel film (Nb2O5-NCF) on niobium foil substrate with facile anodization method."
After thermal treatment in air, the as-fabricated Nb2O5-NCF with pseudo hexagonal phase contains rich oxygen vacancy defects. Scientists found it displayed high electrocatalytic NRR activity with an NH3 yield rate of 2.52×10-10 mol cm-2 s-1 and a FE of 9.81% at -0.4 V in 0.1 M Na2SO4 solution.
During electrocatalytic NRR, the crystalline phase transformation of Nb2O5-NCF results in the electrochromism (EC) phenomenon and the decrease of NRR activity.
The used Nb2O5-NCF electrode can be readily regenerated by low-temperature thermal treatment or applying an anodic potential, recovering high NH3 yield rate and FE with superior recycling stability.
This work was financially supported by the National Key Research and Development Program of China (Grant No.2017YFA0207203) and the Natural Science Foundation of China (Grant No.51872292).
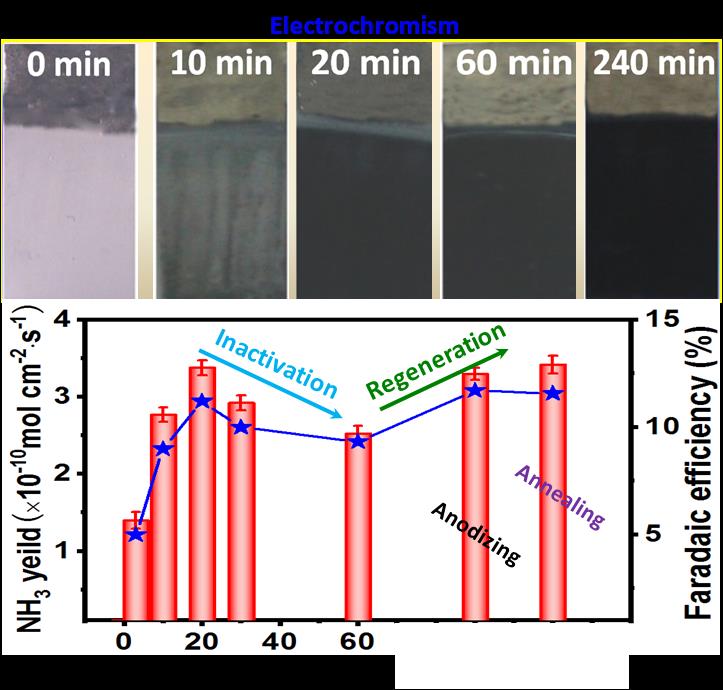
Figure 1. Highly ordered Nb2O5 nanochannel film with rich oxygen vacancies for electrocatalytic N2 reduction: inactivation and regeneration of electrode. (Image by KANG shenghong and WANG Jialu)
Link to the paper: Highly ordered Nb2O5nanochannel film with rich oxygen vacancies for electrocatalytic N2reduction: inactivation and regeneration of electrode
Contact:
ZHAO Weiwei
Hefei Institutes of Physical Science (http://english.hf.cas.cn/)
Email: annyzhao@ipp.ac.cn