
Chlorine atoms (Cl) have higher reactivity than other oxidants (OH radicals, O3, etc.) in the atmosphere. In recent years, researchers found increased concentrations of Cl precursors in the inland regions. The Cl-initiated atmospheric oxidation reactions become more and more important.
Methacrolein (MACR) is the key intermediate of the atmospheric oxidation of biogenic isoprene. The oxidation and degradation of MACR play an essential role in the formation of atmospheric ozone and secondary organic aerosols.
A team of researchers led by Prof. ZHANG Weijun from the Hefei Institutes of Physical Science (HFIPS) of the Chinese Academy of Sciences (CAS), recently investigated Cl-initiated oxidation reactions of MACR under NOx-free conditions, by using a home-made photoionization TOF mass spectrometer complemented with theoretical calculations.
In this study, scientists used a microwave discharge flow tube reactor to study the oxidation reaction of Cl + MACR. The key species such as intermediate radicals and products during the oxidation process were detected online and confirmed in photoionization mass spectra.
The results showed that the reaction of MACR with Cl atoms could generate the C4H5O and C4H6OCl radicals via hydrogen abstraction and the addition of Cl atom to the C=C double bond, respectively.
"This is the first time the transient C4H5O and C4H6OCl radicals were experimentally detected here," said LIN Xiaoxiao, first author of the paper.
She further explained the mechanism. The C4H5O and C4H6OCl radicals could react with O2 to produce the corresponding peroxy radicals C4H5OO2 and C4H6OClO2. Under low NOx conditions, these peroxy radicals would perform bimolecular reactions with themselves and the HO2 radicals.
Combined with theoretical calculations, the resulting specific products could be identified in the photoionization mass spectra.
At the end of the experiment, they measured the time evolutions of products by kinetic experiments and compared it with theoretical model results.
This work elucidated the chemical mechanisms of the Cl-initiated oxidation of MACR, which is helpful to understand the chemical behavior of MACR in the atmosphere.
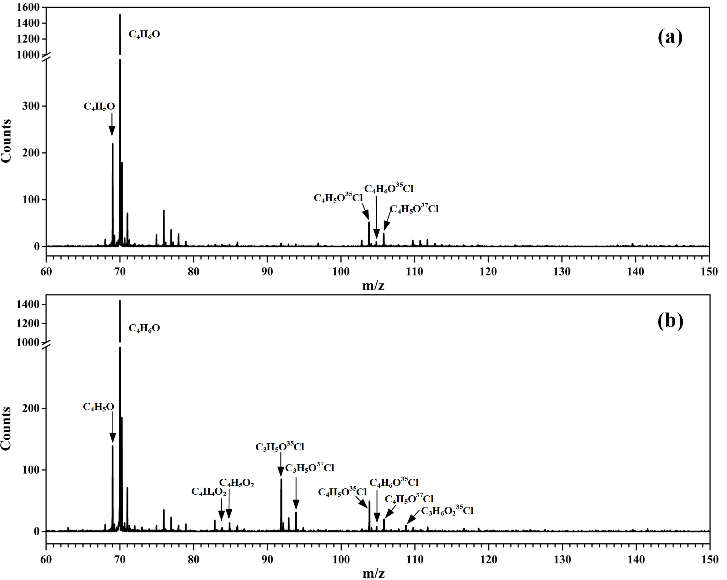
Fig. 1. Photoionization mass spectra acquired (a) without O2 and (b) adding O2 into the fast flow tube. (Image by LIN Xiaoxiao)
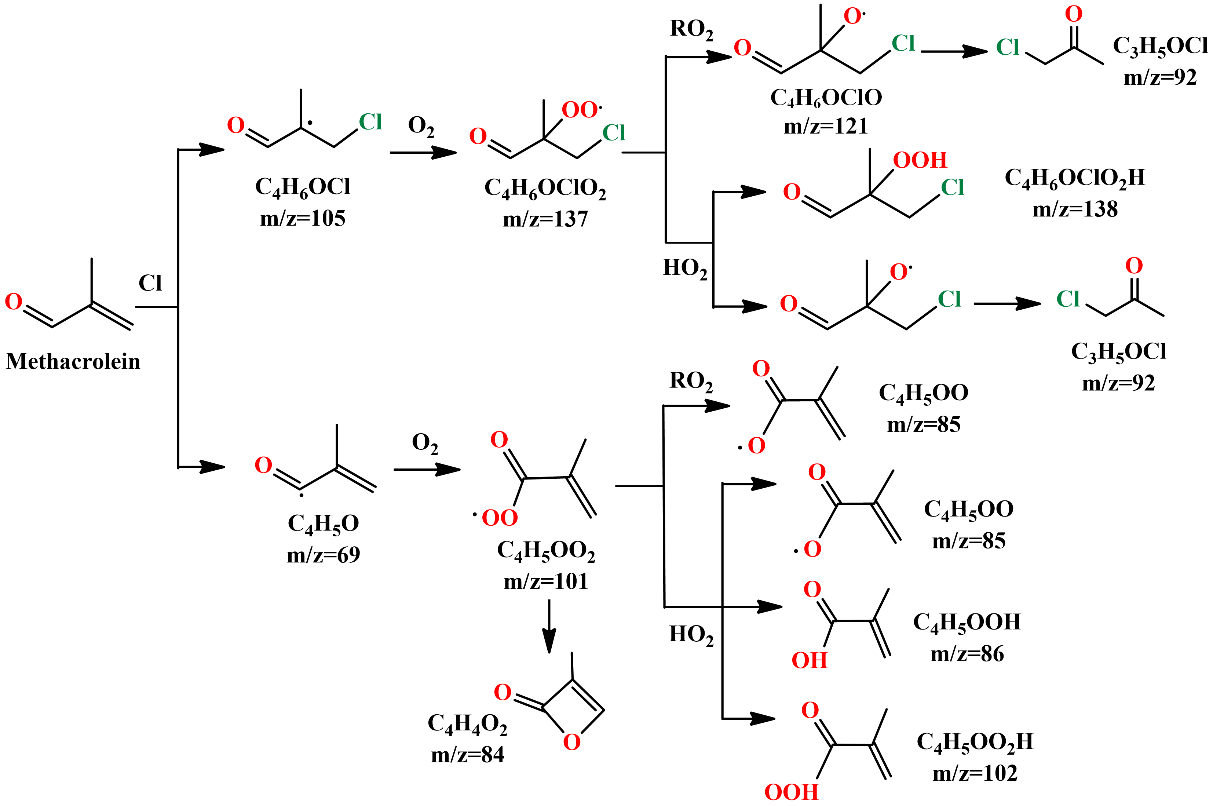
Fig. 2. Reaction mechanisms of Cl-initiated oxidation of MACR under NOx free conditions. (Image by LIN Xiaoxiao)