
Recently, the team from Institute of Solid State Physics, Hefei Institute of Physical Sciences of Chinese Academy of Sciences, made breakthrough in the research of non-metallic heteroatom doping to regulate catalytic performance.
In this work, N and P co-doping carbon-coated cobalt-based catalyst (Co@NPC) was synthesized, and the effect of heteroatom doping in the catalyst on the selective hydrogenation of cinnamaldehyde was explored.
The related research results were published in Journal of Catalysis.
As potential material in the field of catalysis, heterogeneous catalyst of metal nanoparticles supported on non-metallic atoms doped carbon supports demonstrate great catalytic activity in the catalytic hydrogenation reaction. Compared with metal atoms, non-metal atoms possess greater electronegativity. Therefore, the introduction of non-metal heteroatoms can effectively modulate the electronic structure and chemical properties of materials, and produce rich active sites to improve catalytic performance.
In this study, researchers prepared Co@NPC and applied it in the aqueous hydrogenation reaction of cinnamaldehyde. The catalyst was coated by the carbon layer during the synthesis process (hydrothermal and calcination). Once the carbon formed a coating layer, it could prevent the metal from sintering, leaching and growing up. Furthermore, this structure displayed highly dispersed abundant active sites thus contributing to the excellent activity catalyst performance.
It was found that Co@NPC exhibited excellent performance in reaction. 92.1% conversion of cinnamaldehyde and 79.7% selectivity of cinnamaldehyde could be achieved respectively under the reaction conditions of 80 ℃, 0.5 MPa hydrogen pressure and 5 h. Compared with the catalyst Co@C without doping, N, P co-doping can significantly improve the catalytic performance, and control the activity and selectivity of the reaction by adjusting the doping amount.
Their research proved that P doping could improve the selectivity of cinnamyl alcohol, while N doping was closely related to the catalytic activity of the catalyst.
The result of density functional theory (DFT) calculation showed that the electronegativity difference between N, P and C atoms led to the interaction between electrons, changed the ratio of Co and CoO in the catalyst, thereby changing the adsorption strength of cinnamaldehyde on the surface of the catalyst. The reaction products can be adjusted according to the difference of adsorption energy.
This work showed that doping of non-metallic atoms made it more possible to regulate the electronic structure of catalyst, which provided a new strategy for the design of catalyst for selective hydrogenation of biomass derivatives.
This work was supported by the National Natural Science Foundation of China.

(a) Illustration of the preparation process of the Co@NPC; (b), (c) SEM image; (d) TEM image;(e) HRTEM image. (Image by SHEN Yue)
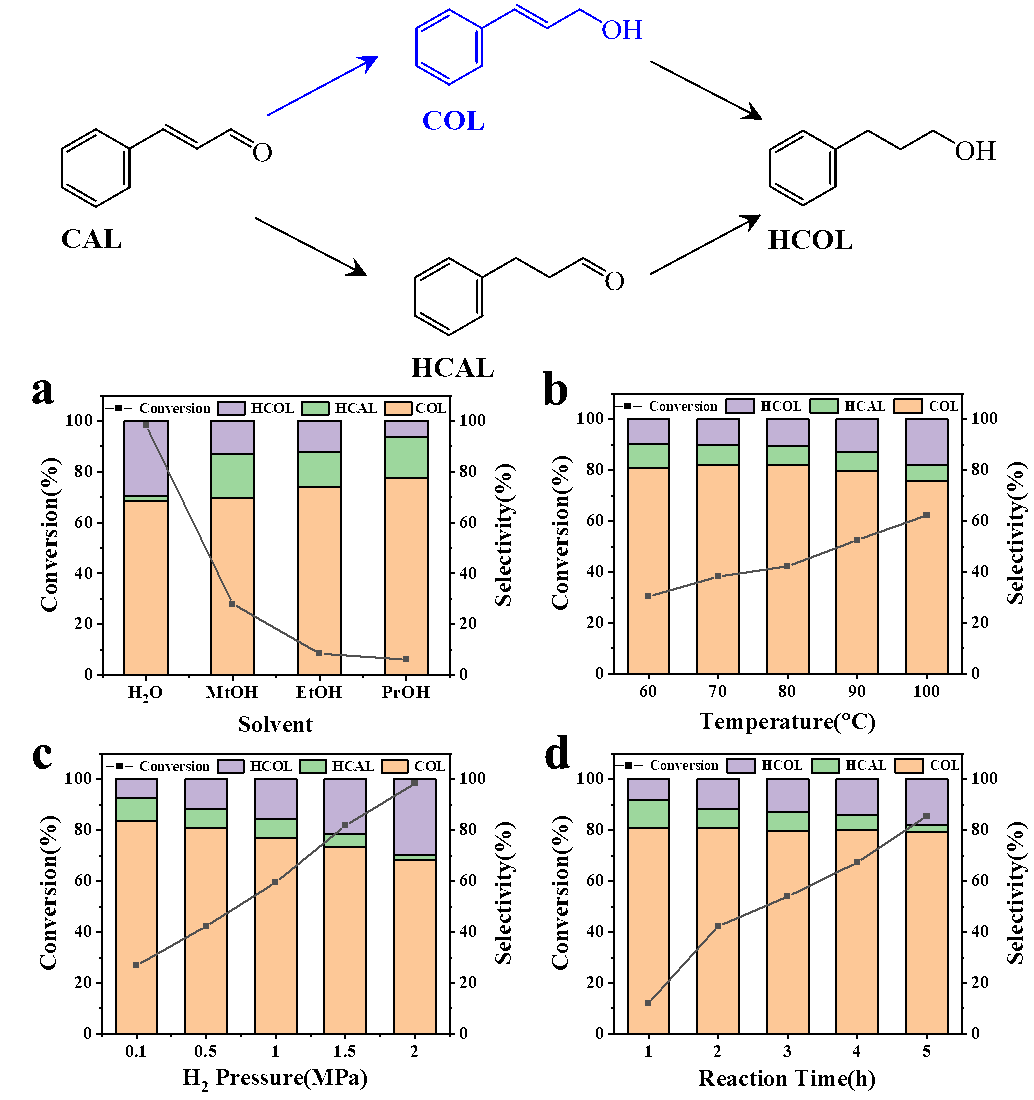
(a) The effect of solvent; (b) The effect of reaction temperature; (c) The effect of H2 pressure; (d) The effect of reaction time. (Image by SHEN Yue)