
According to a research published in Renewables recently, a team led by Prof. HU Linhua. from Hefei Institutes of Physical Science (HFIPS), Chinese Academy of Science (CAS) successfully improved the performance of metal ion batteries enhancing the electric conductivity and ionic mobility of electrode materials.
Anatase TiO2 has attracted tremendous interest as a promising anode for lithium-ion batteries (LIBs) due to its unique features, e.g., stability, low cost, environmentally benignity, and safe. However, as a semiconductor, the intrinsic low ionic and electrical conductivity of TiO2 will cause limited capacity and inferior rate capabilities and cycling performance, seriously hindering its practical applications.
In this study, scientists investigated the properties of oxygen vacancies (VO) and carbon co-dopedO2 hollow spheres (HS-TiO2) and compared them with fully oxidized white TiO2 hollow spheres (W-TiO2).
They found that the bandgap was smaller when carbon dopant and oxygen vacancies (VO) were added to anatase TiO2. Also, the presence of localized electrons led to a lower barrier for Li ions to move, which speeds up the rate of ion diffusion. This made the oxygen vacancies and carbon co-doped TiO2 hollow sphere (HS-TiO2) more reversible than the fully oxidized TiO2 (W-TiO2) when Li ions were added and taken away. By changing the duration of a solvothermal reaction, scientists can control the internal structure of spheres, making them either solid, porous, or hollow. This adjustment allows them to maintain a consistent shape while altering the interior configuration.
The study's findings pave the way for further exploration and optimization of carbon co-doped TiO2 hollow spheres and other similar materials, according to the team.
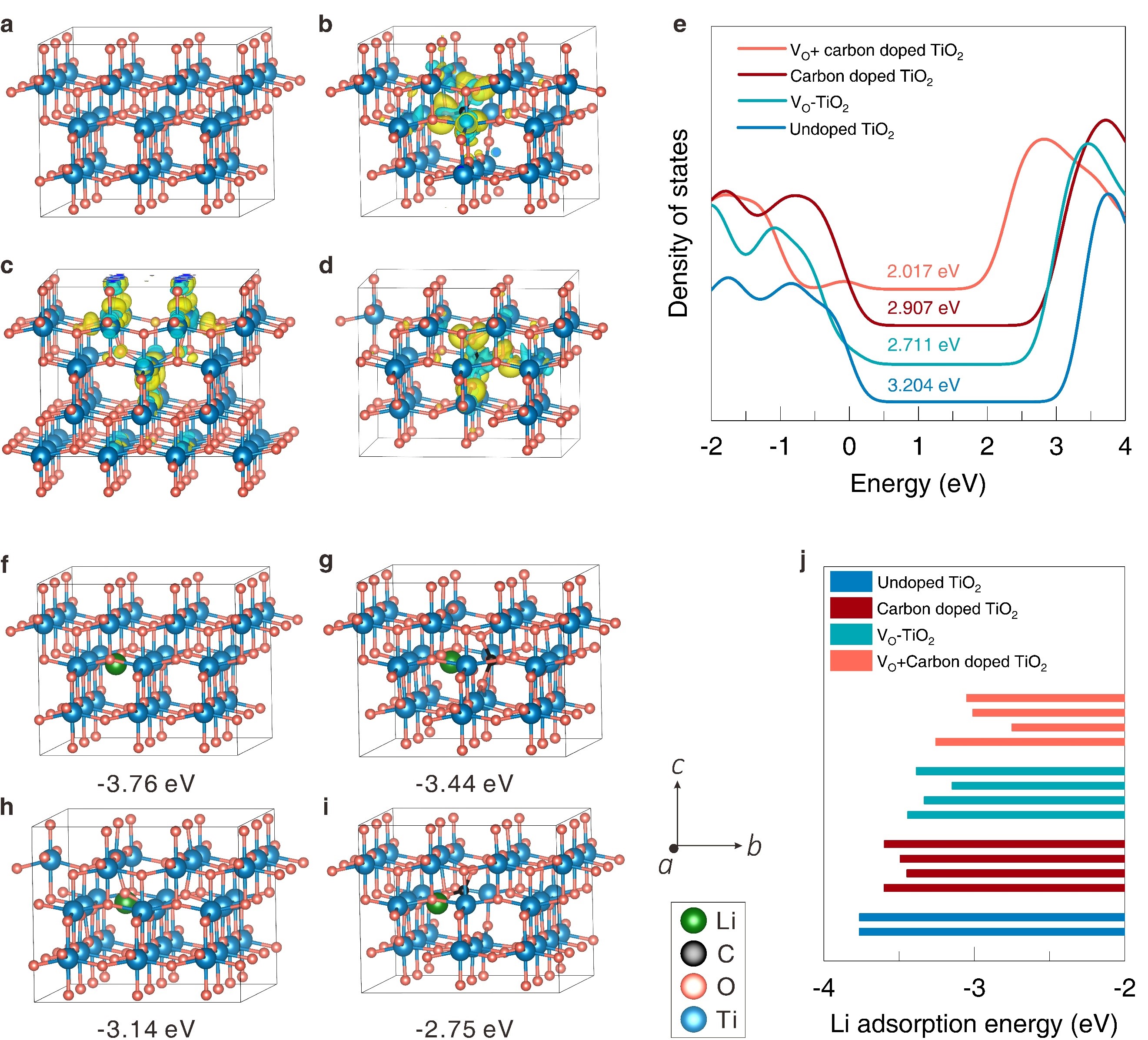
(a-d) Calculated charge difference densities, (e) The total density of states (TDOS) plots, and (f-j) Adsorption energy comparison at the same location site of Li+ of four different location sites of Li+.

(a, b) SEM, (c, d) TEM, (e, f) HRTEM and (g-i) EDS mapping images of HS-TiO2. (Image by LI Zhaoqian)