
Polymeric nitrogen and polynitrogen are believed to be high-energy-density materials. Studies have been conducted to reduce synthesis pressure and improve polymeric nitrogen stability by introducing other elements. However, all patterns of the polymeric nitrogen are negatively charged or covalent bonds are formed between introduced elements and nitrogen atoms by high pressure methods. More research is needed to understand the effects of charge transfer on polymeric nitrogen patterns.
Recently, a team led by Prof. WANG Xianlong and ZENG Zhi studied N-F compounds at high pressures using evolutionary algorithm.
Two new compounds, N6F and NF, were studied, with nitrogen content higher than that of the previously established fluorine-rich compounds NF3 and NF5. Interestingly, a positively charged polymeric nitrogen network is observed in N6F above 180 GPa. The NF compound exists above 120 GPa and becomes metastable at 0 GPa, where nitrogen atoms form zigzag chains. NF has the highest gravimetric energy density of any reported binary covalent nitrogen-compound. The gravimetric and volumetric energy densities of NF are 1.25 and 2.21 times higher than TNT values, respectively, suggesting that it could be a potential candidate for high-energy-density materials.
This work demonstrates how to synthesize new forms of polymeric nitrogen and how to produce a positively charged polymeric nitrogen network. When the introduced element is more electronegative than nitrogen, a positively charged 3D nitrogen network can be realized. It is proposed that the chemical reaction between N2 and F2 under pressure is a new route to the synthesis of polymeric nitrogen, and subsequent experiments are highly anticipated.
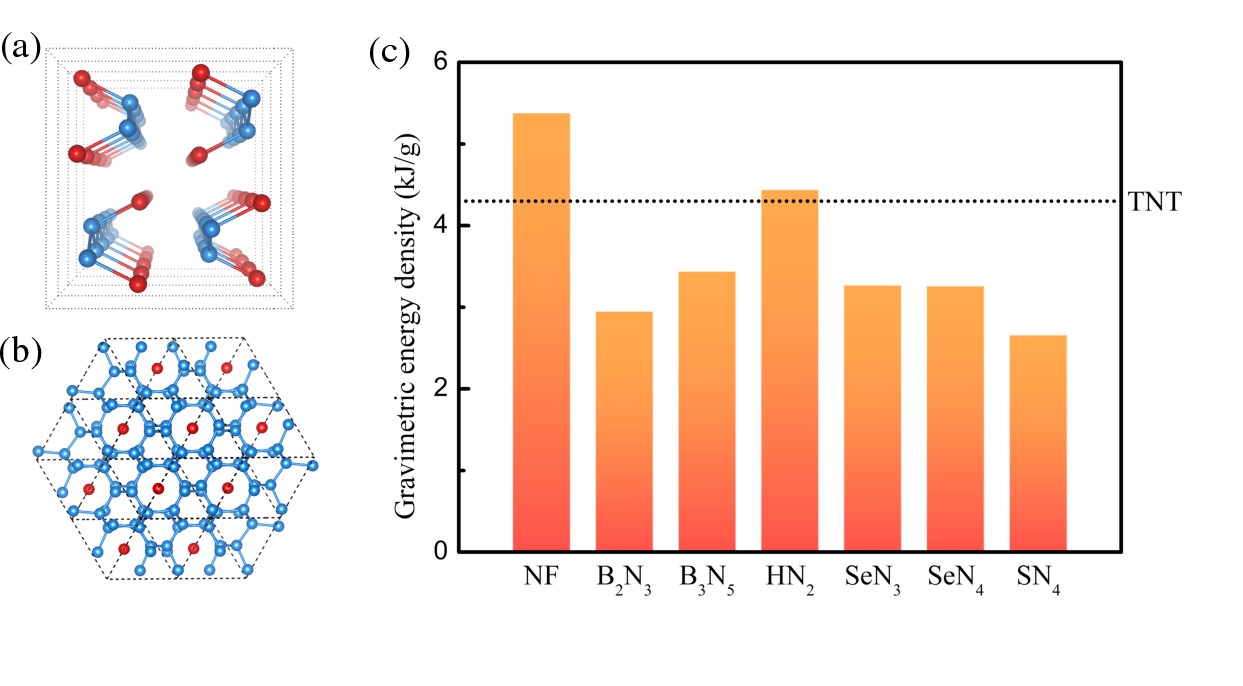
Fig. (a) Crystal structure of the NF compound. (b) Crystal structure of the N6F compound. The blue and red spheres represent nitrogen and fluorine atoms, respectively. (c) Gravimetric energy density of NF and other nitrogen-bearing compounds. The energy density value of TNT is indicated by a dotted line.(Image by ZHANG Jie)