
As a key precursor for the formation of new particles, sulfuric acid (H2SO4) plays an important role in the formation of aerosols and clouds in the atmosphere. Gas-phase sulfuric acid molecules can easily form molecular clusters at the beginning of nucleation through hydrogen bonding and other interactions. Therefore, study on the structure and spectroscopy of H2SO4 and the sulfuric acid–water clusters are of great significance for revealing the nucleation mechanism of new particles.
Recently, a team of researchers obtained the high-resolution slow photoelectron spectrum (SPES) of H2SO4 and observed three electronic states of H2SO4+, X2A, A2A and B2A combined with high-level theoretical results.
This research team was led by Prof. ZHANG Weijun from the Hefei Institutes of Physical Science (HFIPS) of the Chinese Academy of Sciences (CAS), in collaboration with scientists from Synchrotron SOLEIL, France.
"This is the first time the "pure" high-resolution SPES of H2SO4 was found," said Lin, who is a member of the team, "without contaminations from other species in the continuous molecular beam."
In this research, they investigated the vacuum ultraviolet (VUV) photoionization of the sulfuric acid, using the state-of-the-art method of double imaging photoelectron photoion coincidence (i2PEPICO) complemented with theoretical calculations.
The adiabatic ionization energy of the H2SO4 towards the X2A cationic ground state, measured to be 11.684 ± 0.006 eV, matched perfectly with high-accuracy theoretical data in the literature.
When increasing photon energies, the H2SO4+ cation dissociates into HSO3+ and OH fragments. The adiabatic appearance energy of HSO3+ and OH was measured to be 13.498 ± 0.007 eV. In addition, the enthalpies of formation for the species were determined by thermochemical cycle.
This work provides fundamental data and measurement method for in-depth understanding of atmospheric nucleation mechanism.
The findings were published in Physical Chemistry Chemical Physics.
The study was financially supported by the National Natural Science Foundation of China, the International Partnership Program of CAS, and the Key Program of Research and Development of Hefei Science Center of CAS.
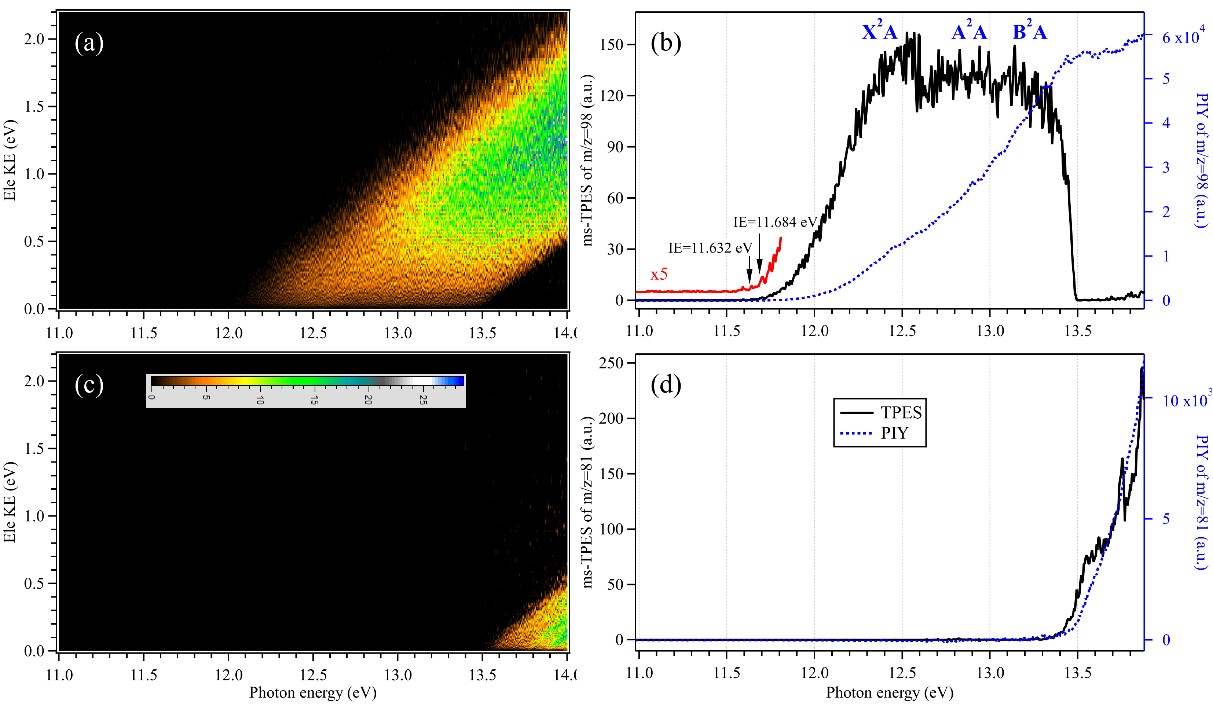
Figure 1. Mass-selected photoelectron matrices, slow photoelectron spectra (SPES, black solid lines) and photoionization yields (PIYs, blue dotted lines). (Image by LIN Xiaoxiao)
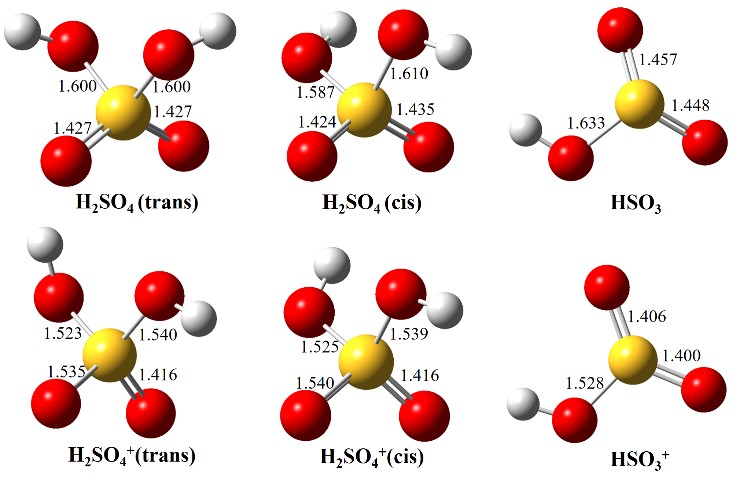
Figure 2. Optimized structures of H2SO4 (trans and cis conformers) and HSO3, as well as their individual cations, in the ground electronic states.(Image by LIN Xiaoxiao)