
The traditional refrigerant represented by Freon is a strong greenhouse gas that leads to serious climate problems. Stimulated by the excellent cooling performance of plastic crystal materials, barocaloric (BC) cooling received much attention recently as a promising green refrigeration technology. However, problems like high driving pressure and low thermal response remain unsolved, which hindered their practical applications.
In a new study, published on Nature Communications, scientists led by Prof. TONG Peng at the Institute of Solid State Physics, Hefei Institutes of Physical Science (HFIPS), Chinese Academy of Sciences (CAS), described how they discovered excellent BC cooling performance in n-alkanes with self-developed BC test platform, which provided a new perspective for developing the application-oriented green refrigeration technology.
"It’s safe and green," explained LIN Jianchao, first author of the paper, “and the cost is low. Current refrigerating exploration tends to use solid-state phase transition materials. Now we have a future with solid-liquid phase transition (L-S-T) materials in mind."
The material they found is called n-alkanes. By applying a low pressure of around 50 Megapascals, a colossal entropy change of ~ 700 J kg-1 K-1 was triggered. This is comparable to those of the commercial Freon-based refrigerants and at least three times larger than the reported values of existing BC materials driven by the same pressure. Aside from that, the adiabatic temperature change driven under this pressure also reaches the highest value of the existing BC materials.
In this paper, they further revealed the possible BC mechanism. The Raman study and Molecular Dynamics Simulation reveal that applying pressure to the liquid state suppresses the twisting and random thermal motions of molecular chains, resulting in a lower configurational entropy. When the pressure is strong enough to drive the L-S-T, the configurational entropy will be fully suppressed and induce the colossal BC effect.
The excellent BC properties, tunable operating temperatures, low-cost raw materials and well-known thermal properties all suggest n-alkanes are promising refrigerants for caloric cooling.
"We expect more excellent BC performance by evoking L-S-T materials in the near future," said LIN.
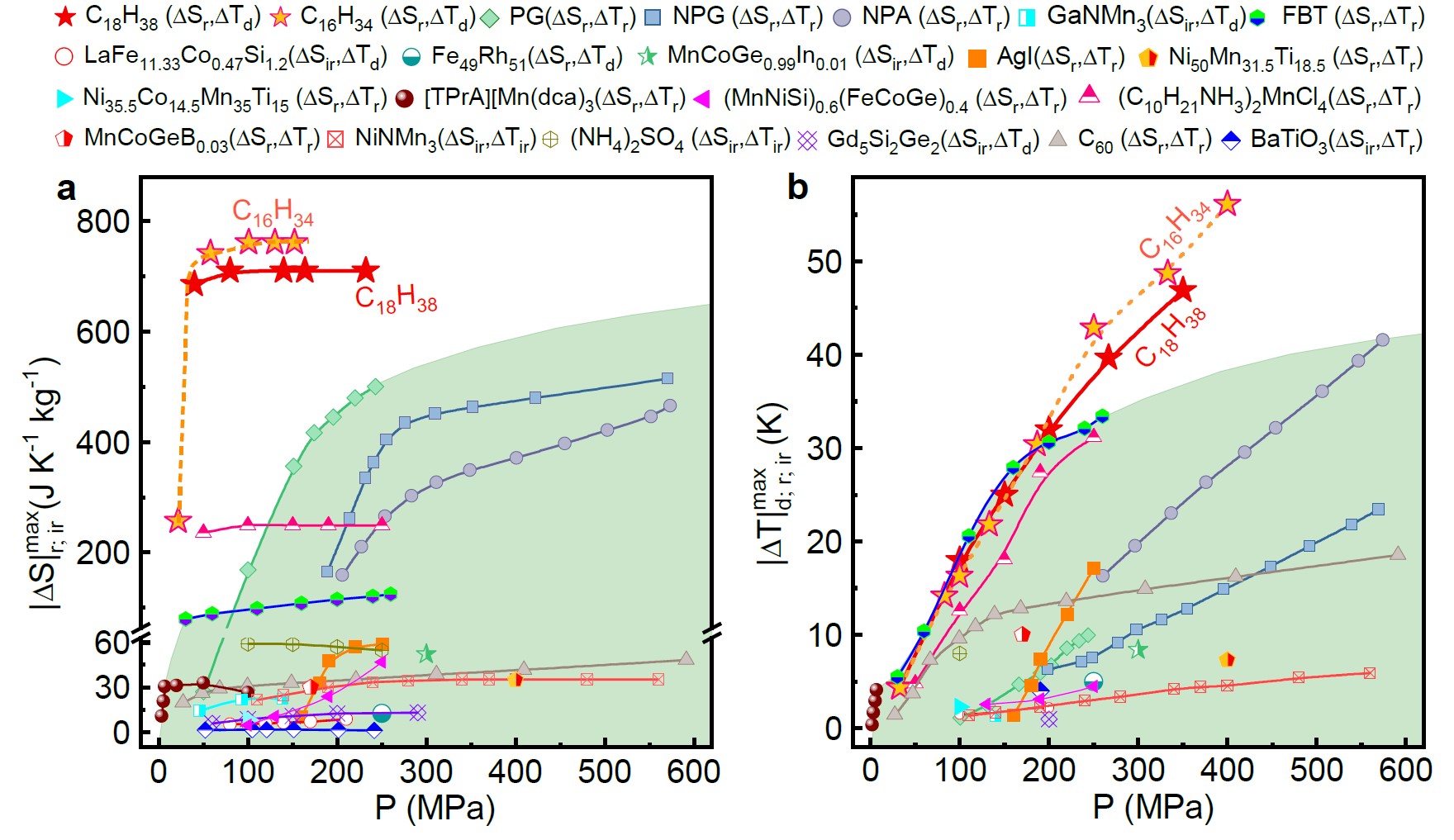
Fig. 1 Comparision of BC performance between CnH2n+2 and the existing BC materials. The maximum adiabatic temperature change (a) and isothermal entropy change (b) as a function of pressure for C18H38 and C16H34, are shown along with the reported BC materials.(Image by LIN Jianchao)
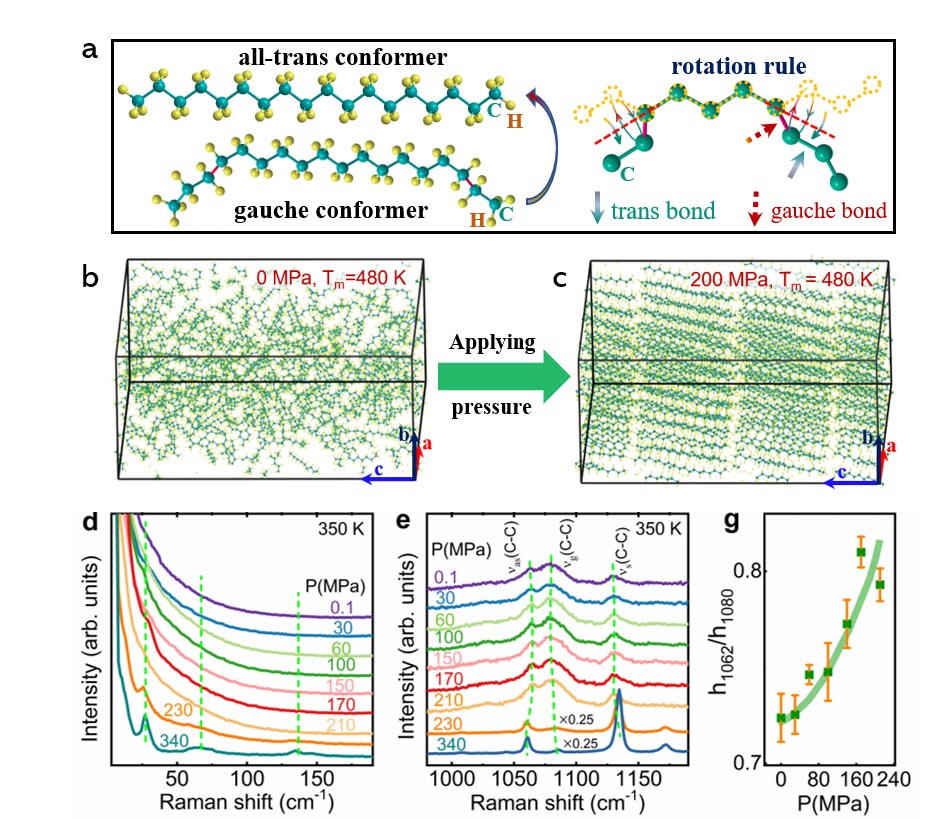
Fig. 2 The barocaloric mechanism revealed by Molecular Dynamics Simulation (a-c) and Raman spectra under different pressure (d-g). (Image by LIN Jianchao)