
Recently, a research team led by Prof. KONG Lingtao from Institute of Solid State Research, Hefei Institutes of Physical Science (HFIPS) of Chinese Academy of Sciences (CAS) proposed a pre-assembly strategy to anchor single atoms on carbon nitride nanosheets. They prepared a series of single atom-loaded carbon nitride Fenton-like catalysts for the degradation of tetracycline pollutants in water, which increased the catalytic activity by 1-2 orders of magnitude.
The research results were published in Separation and Purification Technology.
The Fenton-like process was radical dominated reaction. H2O2 and peroxymonosulfate (PMS) are two commonly used Fenton-like oxidants, and reducing the reactive species' migratory distance to the pollutant molecules is desirable for performance enhancement due to the reactive species produced from H2O2 and PMS activation have a very short half-life period.
Single-atom materials have better activation effect on oxidants. Owing to the arranged nanosheets structure, tunable surface area, benign biocompatibility and high stability in harsh environments, graphitic carbon nitride (CN) can serve as a promising support material for radicals confinement. At the same time, its abundant nitrogen can provide ideal sites for the embedding of metal ions, forming unique coordination structures and electronic configurations. Therefore, the immobilisation of metal atoms on carbon nitride nanosheets can confine the free radicals near the contaminants, and effectively improve the Fenton-like catalytic efficiency.
In this research, researchers proposed a pyrolytic coordination polymerization pre-assembly strategy with broad-spectrum versatility. They anchored single atoms on carbon nitride nanosheets and demonstrated versatility in Fenton-like catalysis.
As a proof of concept, the SA-Cu-CN was chosen as a model material for the degradation of tetracycline (TC) and mechanistic elaboration. The Fenton-like catalytic activity of SA-Cu-CN could reach one to two orders of magnitude higher than that of the control materials.
EPR analysis and quenching experiments showed that the catalytic system in which hydroxyl radical and sulfate radical generation played a crucial role in the degradation of TC. Combining UPLC-MS analysis with DFT theoretical calculations, the degradation pathways and product toxicity of TC were analyzed and characterized. The SA-Cu-CN Fenton-like catalyst showed great ability to treat organic pollutants in depth.
In addition, a series of single-atom catalysts such as SA-Fe-CN, SA-Co-CN and SA-Mn-CN were synthesized by the same preparation method. They all showed good Fenton-like catalytic activity.
This study is of great importance for the development of Fenton-like catalysts and their applications in water treatment, according to YANG Dandan, first author of the paper.
The above work was supported by the National Key R&D Program, the National Natural Science Foundation of China, the Natural Science Foundation of Anhui Province and the President's Fund of the Hefei Research Institute.
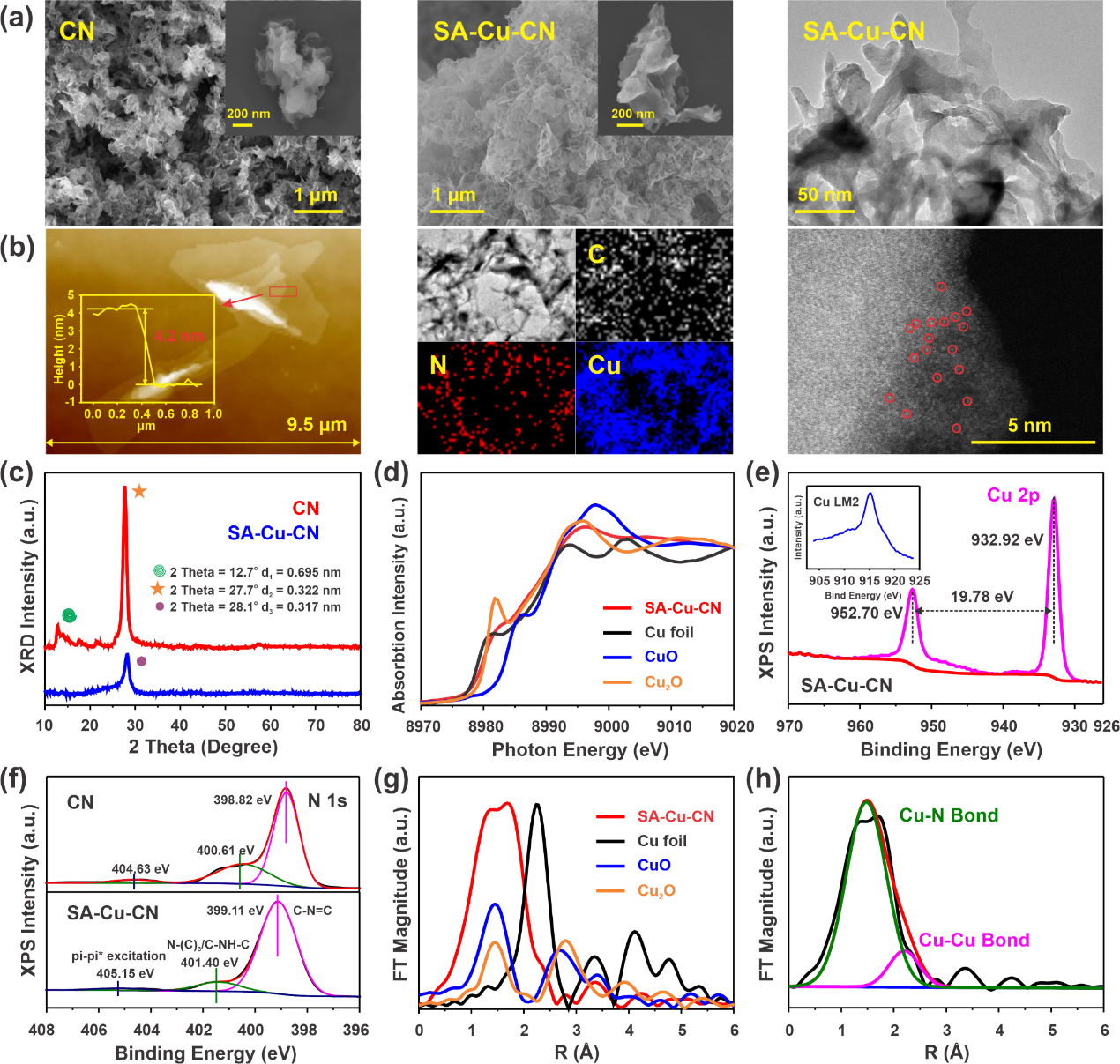
Figure 1. Morphological and structural characterisation of CN and SA-Cu-CN. (Image by YANG Dandan)
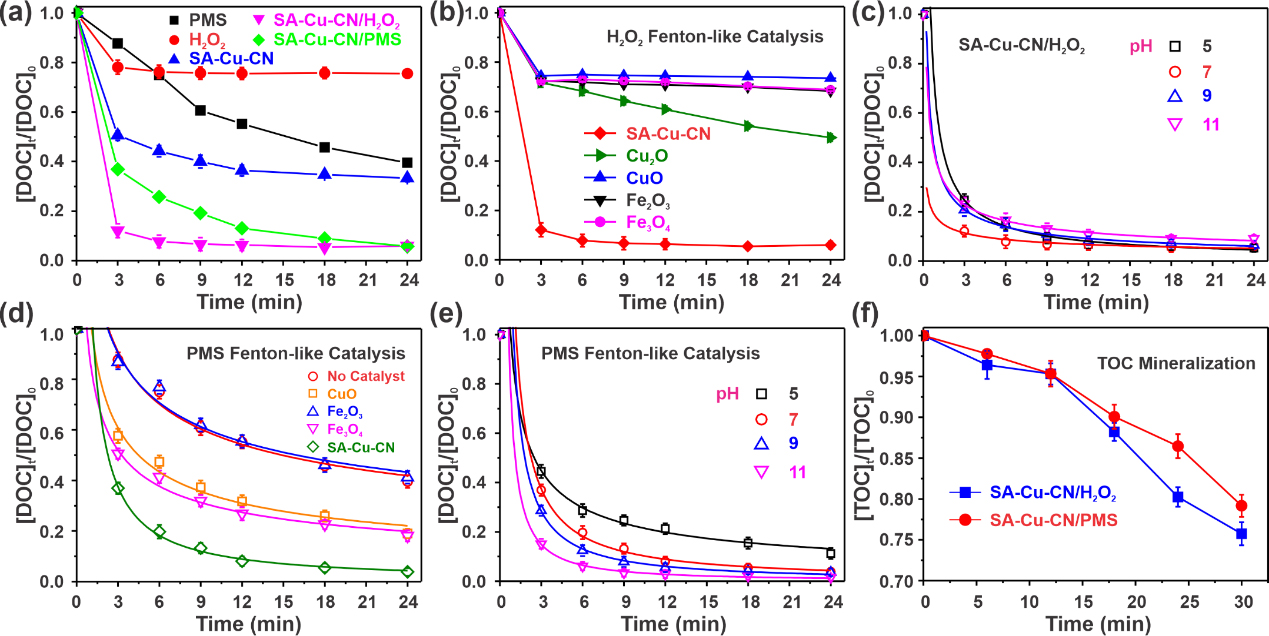
Figure 2. Exploration of Fenton-like catalytic performance of SA-Cu-CN. (Image by YANG Dandan)
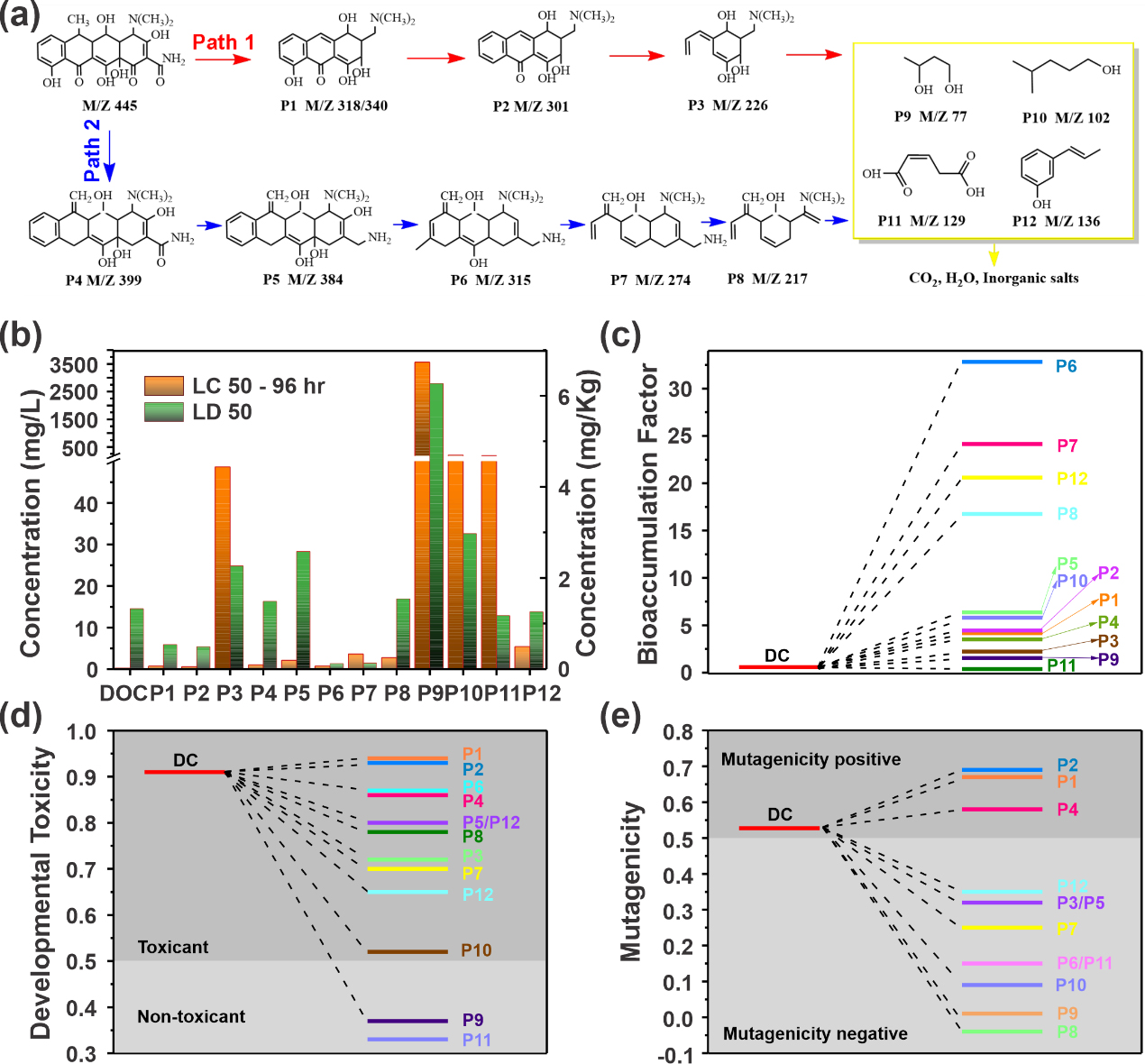
Figure 3. Pathway analysis of tetracycline degradation and identification and toxicity assessment of its products. (Image by YANG Dandan)