
Researchers Reveal Molecular Basis of Photo-State Recognition in MagRed System
A research team led by Prof. WANG Junfeng and Associate Prof. ZHU Lei from the High Magnetic Field Laboratory, the Hefei Institutes of Physical Science of the Chinese Academy of Sciences, has uncovered the structural basis by which the affibody Aff6 specifically recognizes different photoexcitation states of the photosensitive protein DrBphP in the red/far-red optogenetic system MagRed.
The study was published in Journal of Photochemistry and Photobiology B: Biology.
Optogenetics uses light to precisely control biological processes. Infrared-based systems such as MagRed are particularly suitable for deep tissue applications because of their strong penetration and low phototoxicity. MagRed consists of the photosensitive protein DrBphP and its binding partner Aff6. However, how Aff6 distinguishes between different light-induced states of DrBphP has remained unclear, limiting further optimization of the system.
In this study, the researchers used surface plasmon resonance to demonstrate that the monomeric photosensory core module of DrBphP alone is sufficient to drive light-induced heterodimerization with Aff6. Notably, they found an approximately 23-fold difference in binding affinity between the Pfr and Pr states.
They then employed nuclear magnetic resonance titration and chemical shift perturbation analysis to map the interaction interface. The results showed that Aff6 primarily binds to the phytochrome-specific domain and the C-terminal region of the helical spine of DrBphP. Further molecular docking and site-directed mutagenesis revealed that key aromatic interactions play a central role in this state-specific recognition.
Additionally, the study showed that Aff6 binding stabilizes the Pfr state through an allosteric effect, thereby slowing its dark reversion to the Pr state.
This work provides guidance for developing more precise and efficient red/far-red optogenetic tools, according to the team.
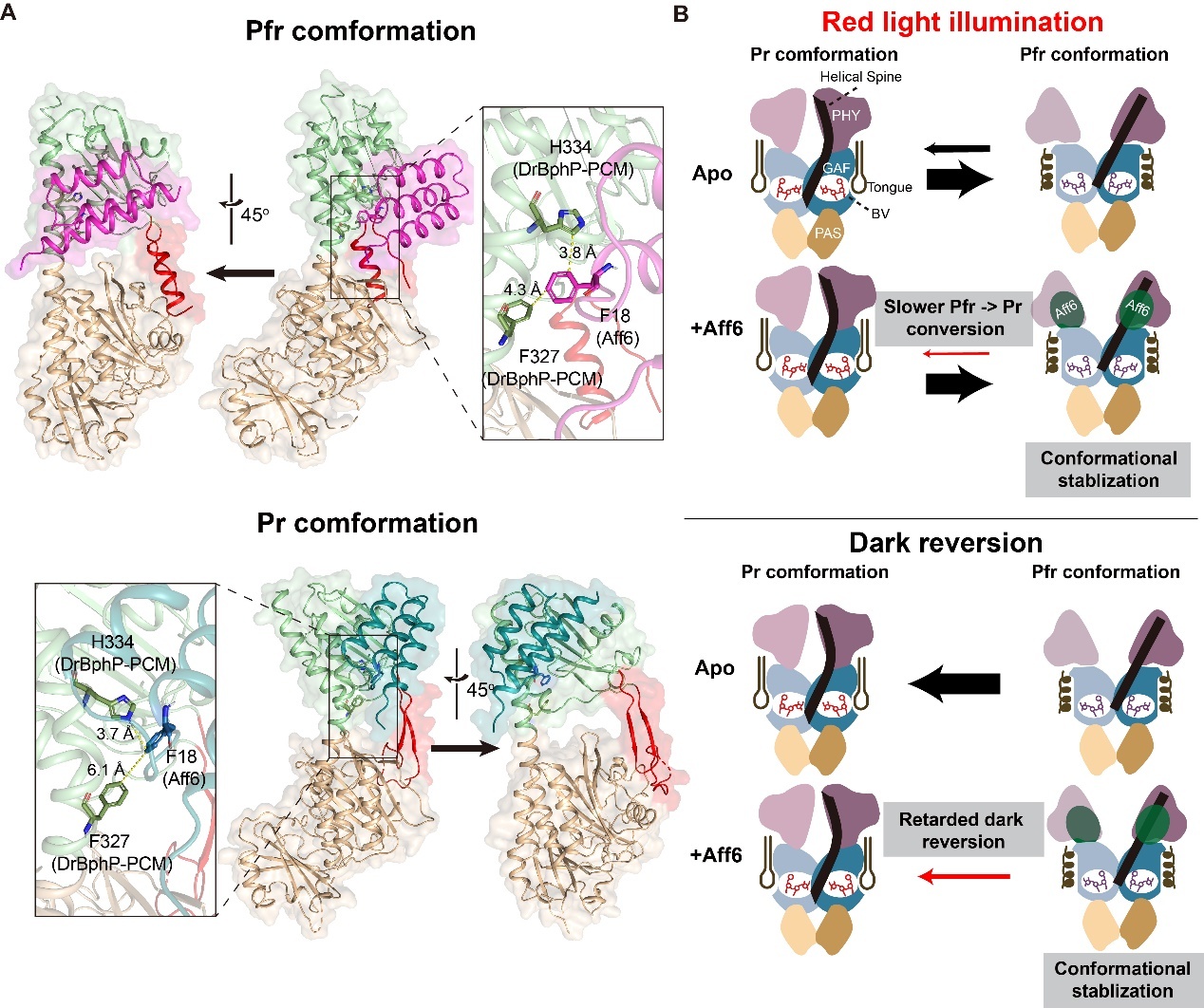
Molecular mechanism of the photo-state-dependent interaction between DrBphP and Aff6. (Image by ZHU Lei)