
Tetracycline (TC) is one of the most common broad-spectrum antibiotics. However, typically less than 30% of the antibiotic dose was absorbed by human and animal, while the remainder released into the environment through urine. The coexistence of TC and water hardness ions (Ca2+ and Mg2+) in natural water is a widespread pollution phenomenon. Especially, TC and water hardness ions could easily form TC-metal complex pollution in the environment.
In a recent study published on Journal of Cleaner Production, scientists at Institute of Solid State Physics, Hefei Institutes of Physical Science (HFIPS) of the Chinese Academy of Sciences used capacitive deionization (CDI) technology to simultaneously remove TC and water hardness ions (Ca2+ and Mg2+) from water based on unique innate advantages of CDI technology. The method is efficient and eco-friendly with low-cost.
The team derived hierarchical porous carbon material from kelp and used it as symmetric electrodes. A maximum capacity of 925.3 mg g-1 for TC removal was achieved, which is much higher than capacity of other materials reported so far.
Besides, the removal efficiency was as high as over 90% even after five CDI adsorption-desorption cycles. Meanwhile, the metal ions with opposite charges were adsorbed on the counter electrode with a preferential ion electrosorption performances, proving the effective CDI technology for water purification.
This work has paved a way to synchronously eliminate multiple organic and inorganic contaminants from water by using CDI technology.
Link to paper: Synchronous removal of tetracycline and water hardness ions by capacitive deionization
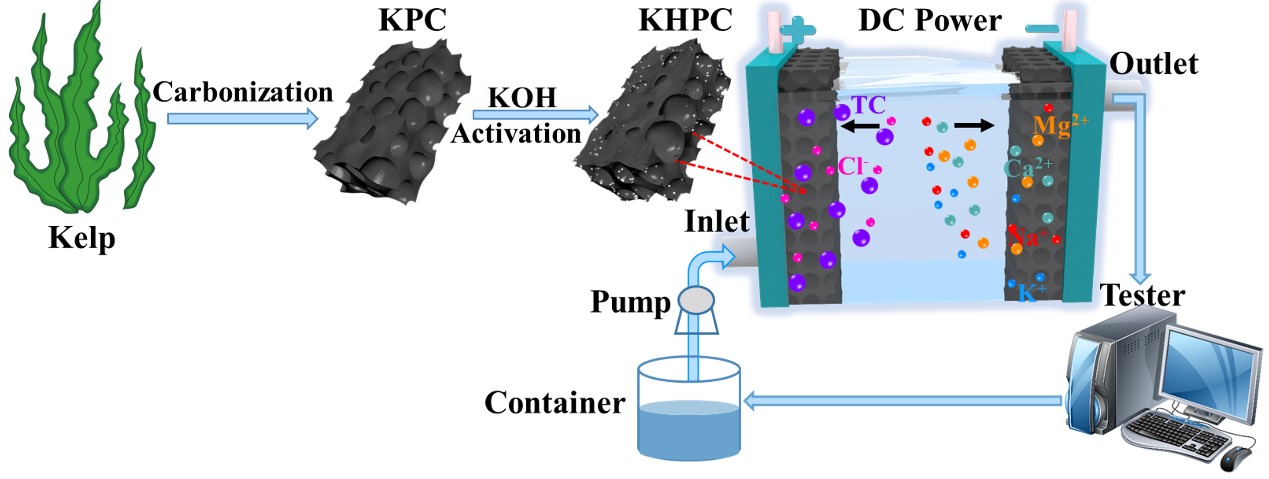
Schematic diagram for synchronous removal of tetracycline and water hardness ions by capacitive deionization (Image by ZHOU Hongjian)
Contact:
ZHAO Weiwei
Hefei Institutes of Physical Science (http://english.hf.cas.cn/)
Email: annyzhao@ipp.ac.cn